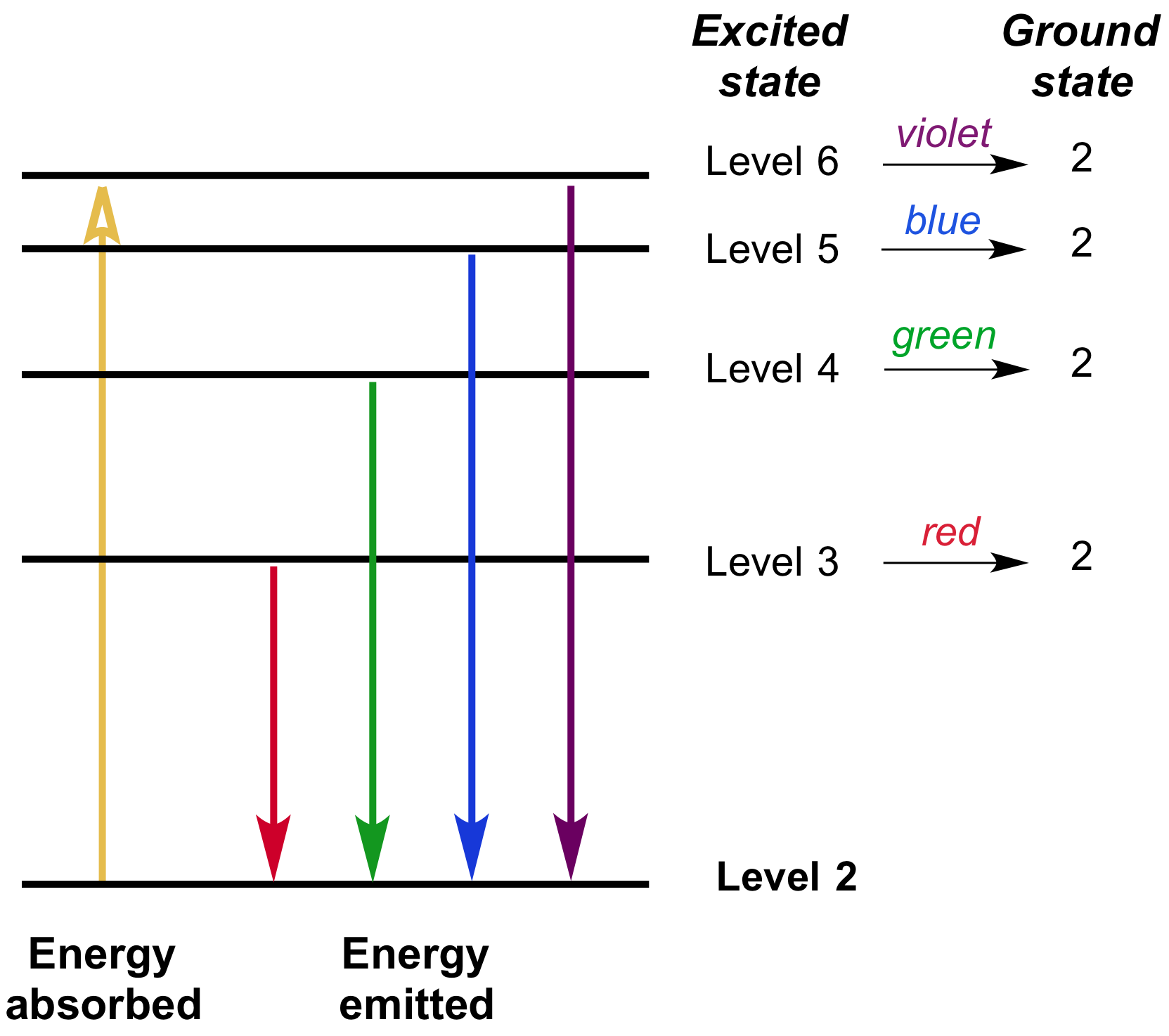
When electrons return to a lower energy level, they emit energy in the form of light. The light emitted is composed of discrete colors. Below are images of the spectra of the lines of each element which would be observed as emission lines in an electrical gas discharge. Atomic emission spectra are produced when excited electrons return to the ground state. This is an attempt to give a reasonable accurate picture of the appearance of the neon spectrum, but both the images are composite images. Each colour band corresponds to a specific energy of photon released by electrons jumping. Main idea: put a large voltage across the gas. The spectra of neon gas as observed through a diffraction grating. The principle of the atomic emission spectrum explains the varied colors in neon signs, as well as chemical flame test results (described below). 
(b) The energy of the orbit becomes increasingly less negative with increasing n.ĭuring the Nazi occupation of Denmark in World War II, Bohr escaped to the United States, where he became associated with the Atomic Energy Project. colors, called emission spectrum, depending on its composition. Newton classified this spectrum in a range of seven different colors (Red, Orange, Yellow, Green, Bleu, Indigo and Violet. Click here to get an answer to your question The red light of neon signs has a wavelength of 693 nm. \) for a summary).\): The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. In the 1670s, Isaac Newton, during optical experiments observed that a beam of white light was decomposed in a continuum spectrum of all visible colors, like a rainbow, when it pass through a prism.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
